Scratching the Surface of Target Product Profile (TPP)
What is a Target Product Profile and how to create one?
Note: This article was written in collaboration with Arun Kejariwal, a Vibe Bio community member and pharmaceutical industry veteran. We hope you find this explainer article useful and valuable. Share your feedback in the comments section.
Target Product Profile: A Blueprint for Developing Successful Products
Developing a new pharmaceutical drug is a lengthy and expensive process, where research and development can take many years or even decades to complete. To ensure that resources are used effectively and efficiently, companies need to have a clear understanding of what they are trying to achieve with each new drug candidate they develop. This is where the Target Product Profile (TPP) comes in. A TPP is a strategic document that outlines the desired specifications and requirements for a drug to address unmet needs that are expected to exist when the product launches in the market. In this blog post, we will explore the importance of TPP and its creation process.
What is the Purpose of the TPP?
The main purpose of the TPP is to guide the drug development team in their efforts to create a product that meets the needs of patients, healthcare providers, payers, and other stakeholders. TPP is often referred to as the "north star" for the research, development, and commercialization team. It ensures that all stakeholders are aligned on the goals and expectations for the product. The progress and success of the product being developed is measured against the attributes defined in the TPP at every stage of the development, and any deviations from the profile require a reassessment of the product's viability and continued investment.
In addition to its internal use, the TPP is a critical communication tool for external stakeholders, including potential investors. The TPP document is well known to investors in the pharma industry and having a TPP available for investment discussions helps ensure clear communication on the company's goals and expectations for the drug.
The TPP is also a critical document for discussion with regulators throughout the development of the drug, from pre-clinical to commercialization. A version of the TPP is included with the New Drug Application (NDA) or Biologics License Application (BLA) submitted to the regulatory agency.
(source: https://downloads.regulations.gov/FDA-2007-D-0256-0003/attachment_1.pdf) (reading: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5478920/)
Path to Creating a TPP
TPP starts as a "product concept" that provides a high-level strategic direction to the discovery/early research team. It outlines what their research project should be able to address at a high level, such as whether it is a disease-modifying or symptomatic therapy, the level of efficacy compared to placebo or standard of care, and the route of administration etc. As the program completes the discovery/early research stage, the "product concept" is further refined and converted to a TPP with specific targets for each attribute.
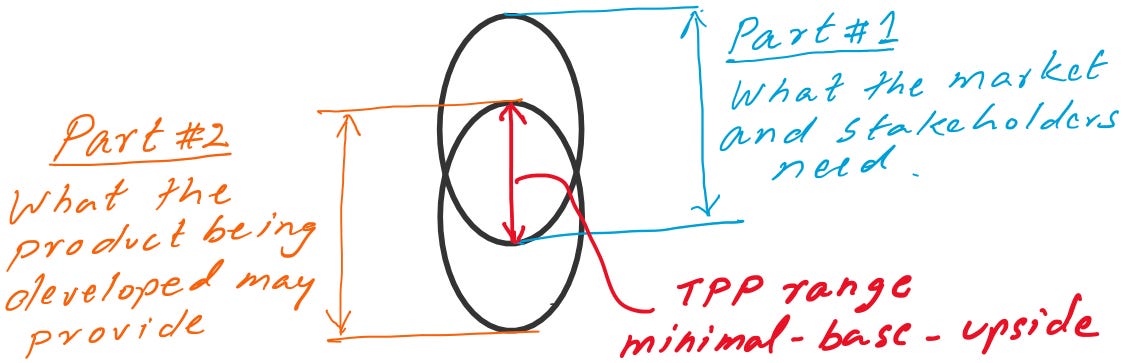
Creating a TPP involves two broad components.
Part I: The first part involves identifying the unmet needs of the market and stakeholders, including patients, healthcare providers, payers, hospitals, etc. This also includes understanding of the commercial potential (size of the target market, competitive landscape, differentiation strategy, pricing, reimbursement considerations etc.) of addressing the unmet need.
Part II: The second part involves assessing how much of the unmet need the product being developed can address in a certain time frame. It is our best assessment of the potential scientific strength of the product being developed.
A successful TPP is a balance of these two components and should include adequate aspects of stakeholder unmet needs to be a meaningful product that brings societal benefits and is commercially viable for the developer. Typically, TPP is summarized in one or two pages.
Necessary Attributes in a TPP
Below are some necessary attributes to include in a TPP:
Target indication: The target indication defines the medical condition or disease that the drug aims to treat. It should be clearly defined, and the rationale for selecting this indication should be explained.
Target patient population: The target patient population is the specific group of patients who are expected to benefit from a particular drug or medical product. This includes patients who have a specific medical condition or disease, as well as other demographic characteristics such as age, gender, and ethnicity.
Efficacy: Efficacy refers to the drug's ability to achieve the desired therapeutic effect. It is usually measured by clinical endpoints, such as the reduction in symptoms or disease progression. The TPP should define the level of efficacy required for the drug to be considered successful.
Safety: TPP should define the acceptable safety profile for the drug. This includes the types and frequency of adverse events, as well as any potential drug interactions.
Dosing and administration: The TPP should define the optimal dosing regimen and route of administration for the drug. This includes considerations such as patient compliance, convenience, and the availability of suitable delivery devices.
Considering that the TPP is first drafted well before the actual start of the drug development and, as we are aware that the drug development can take a long time, the TPP needs to be future looking and factor in the likely evolution of the market and associated unmet needs. As such, it also needs to be a living document that evolves with the product development and is routinely refreshed to account for new internal or external information and market changes.
Conclusion
In conclusion, a TPP is a critical tool that guides the product development process in the pharmaceutical industry. It ensures that all stakeholders are aligned on the goals and expectations for the product and helps measure the progress and success of the product at every stage of development. Creating a TPP involves identifying the unmet needs of the market and stakeholders and determining how much of the unmet need the product can address in a certain timeframe. A balance between these two components is essential to ensure that the product is commercially viable and brings societal benefits.
TPP Examples
TPP Example #1 | COVID-19 vaccines
TPP Example #2 Pain associated with diabetic neuropathy